Properties of Water

What's up, world? I'm back! As you can see, the UNIT #2 tab is all about water. A couple days ago, our class took notes and did some experiments. I will be putting down all the data we found out about and the notes we took. I'll first do the notes, then the experiments. I'll see you soon, make sure to read it all, ok? Peace!
What is Water?
Water is made up of two hydrogen atoms and one oxygen atom (H2O). An atom is the smallest unit of matter that retains its properties. Atoms consists of protons, electrons, and neutrons. Atoms are usually neutrally charged because the number of protons and electrons are even. When two hydrogen atoms and one oxygen atom combine to form a water molecule, each hydrogen atom shares its single electron with the oxygen, and the oxygen atom shares one of its electrons with the hydrogen atom.
Protons: have a positive charge
Electrons: have a negative charge
Neutrons: have no charge (neutral)
Ions: unevenly charged atoms
Protons: have a positive charge
Electrons: have a negative charge
Neutrons: have no charge (neutral)
Ions: unevenly charged atoms
Water is a Polar Molecule

Shared electrons spend more time around the oxygen atom than the hydrogen atom. The hydrogen atom carries a slightly positive charge while the oxygen atom carries a slightly negative charge. In other words, the oxygen atom is negative and the hydrogen atom is positive. Because of that, the oxygen atom and the hydrogen atoms are attracted to each other.
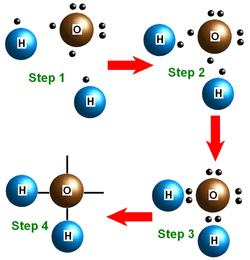
Hydrogen Bonding is a weak attraction between the weakly positive hydrogen atoms and the weakly negative oxygen atoms of a different molecule. Hydrogen bonding is responsible for the unique properties of water. Basically, they all gather together to make water.
Water is a Universal Solvent
Polarity of water gives it the ability to dissolve most compounds. The positive hydrogen side of water is attracted to the negative parts of the compound it is dissolving. Negative oxygen is attracted to the positive parts. This allows water to dissolve and break apart ionically charged substances. Cohesion is when water is attracted to other water molecules. This helps trees pull water high up into their branches. Water is able to pull itself into a shape with the smallest amount of surface area. Surface tension is the attraction of water molecules to themselves helps to create a "film" on the surface of bodies of water. Small critters can "walk" across the water. Adhesion is when water is attracted to other materials.
States of Matter
Water exists in 3 phases: Liquid (water), Solid (ice), and Gas (steam/water vapor). Temperature of a substance is a measure of the kinetic energy of the atoms and molecules which is measured in degrees (Celsius- science), Fahrenheit in US. Heat is a measure of the total kinetic energy of the atoms and molecules which are measured in calories. One calorie is equal to the amount of heat needed to raise the temperature of 1 gram of by 1 degree Celsius. Here is an example: below are two pictures. One picture is a pool and the other is a boiling pot. Here are some questions you can answer.
1. Which object has more heat?
2. Which object has a higher temperature?
1. Which object has more heat?
2. Which object has a higher temperature?
Answers to the questions:
1. The pool is bigger, so it has more heat.
2. Because it is boiling, the pot has a higher temperature
1. The pool is bigger, so it has more heat.
2. Because it is boiling, the pot has a higher temperature
Effects of Heat on Water
High heat of vaporization happens when a hydrogen bond must be broken, so water can absorb a lot of heat without changing phase. It evaporates slowly causing the cooling effect when you sweat. Water has to boil at a high temperature. Most of the water on Earth is a liquid which is good for the oceans. High latent heat of fusion happens when a lot of heat is released when water freezes and a lot of heat is absorbed when ice melts. Water temperature is more stable than air temperature and water moderates climates. When water has a specific heat, it can absorb high specific heat of water which helps regulate the rate at which air changes temperature, which is why the temperature change of seasons is so gradual, not sudden, especially near large bodies of water like the oceans.
What is density? Density is when the physical property of a substance that describes the degree of compactness of a substance or how closely packed together the atoms are. The more closely packed the atoms are in a substance, the more dense it is.
Density= mass/volume
How much stuff in how much space?
Usually substances becomes more dense (more compact) when they solidify. Water is different. It actually becomes less dense in its solid form. Water is the substance able to do this.
What is density? Density is when the physical property of a substance that describes the degree of compactness of a substance or how closely packed together the atoms are. The more closely packed the atoms are in a substance, the more dense it is.
Density= mass/volume
How much stuff in how much space?
Usually substances becomes more dense (more compact) when they solidify. Water is different. It actually becomes less dense in its solid form. Water is the substance able to do this.
Investigating Water Lab
I'm finally finished with the notes! Yes! Now it is time for me to continue on with the experiments that we did in class. I apologize, there are some experiments that I don't have pictures for. However, do you know what is the best part of these experiments? You can actually try this at home as well. Just make you clean up afterwards, ok? Now let's start with the first one, shall we? See you later!
Evaporative Cooling: Station #1 Water and Alcohol
We did an experiment to find out which was cooler on the back of your hand. Each person in our group put one drop of water and one drop of rubbing alcohol on their hand. When we all tested it out, we all agreed that the rubbing alcohol was cooler. To make sure, we blew our hand with the water and rubbing alcohol on it. The alcohol felt cooler when we blew on it. In order to get rid of excess heat, we sweat, everyone knows that right? The sweat (water) absorbs the heat from your body, taking your heat energy with it. This is what you call the effects of heat on water. The reason why sweat is water and not alcohol-based is because it could take away too much of your heat energy.
Water Tension/Cohesion: Station #2 Water on a Penny

We had to decide how many drops of water would fit on a penny before it overflowed. I predicted that it would carry 5 drops of water before it overflows. Man, was I wrong! Get a dropper and fill it with water. Now, slowly drop one drop of water on to a penny every three seconds. Watch the side of the penny to see what it looks like. As we looked at it from the side, it looked like there was a bubble on the penny. We got 32 drops of water onto one penny before it collapsed. How could it hold so much water? The answer is cohesion. Cohesion is when water molecules stick to each other. Because of that, it was able to hold a lot of water on a small penny.
Surface Tension: Station #3 Water on Wax Paper

I want to say right now that we didn't have any wax paper, so we had to use parafilm. Our teacher said that it was similar to wax paper, so we should be fine. Anyways, get a dropper and fill it with water. Get some parafilm and place it on the table. Put some water onto the parafilm and see what happens. The parafilm didn't absorb the water, it just sat there in a bubble-like form. When the water doesn't sink into something, that is because there is surface tension. Surface tension is when the water molecules attract each other to create a "film" on the surface of any bodies of water. Because of that, the parafilm didn't absorb the water.
Surface Tension: Station #4 Paper Clips on the Surface of a Cup of Water

Have you ever tried to make something float on water? We had to make a paper clip float on water with some tweezers. Remember from Station #3, we talked about surface tension. Here's a reminder of what surface tension is. Surface tension is when the water molecules attract each other to create a "film" on the surface of any bodies of water. This experiment is an example of what surface is like. We were able to make 3 paper clips float. We weren't able to do this because then we would have to clean up, but if you put a drop of dish-soap into the water, the paper clips would fall. Do you know why? If soap were to touch the water, the surface tension would dissolve and the paper clips would fall to the bottom of the cup.
Adhesion: Station #5 Graduated Cylinder

Have you ever wondered why water looks different in different cup sizes? The answer to that is adhesion. Adhesion is when water is attracted to other materials. It can also reshape itself when put into something. An example is two different sized cups, but same amount of water in both cups. They will obviously look different. On the graduated cylinder, you can see that there are lines on the cylinder. Why are the lines not straight on the cylinder? That is because the cylinder is curved, when you want to know how much water in the cylinder, you look at the meniscus (where the line is straight). Adhesion is important because living systems are able to survive more when they have other materials around them.
Capillary Action: Station #6 Rolled Up Paper Towel

In this experiment, we placed one end of a paper towel into a beaker filled with water and blue food coloring. We then placed the other end into a beaker with nothing. In a couple of days, the colored water will be transferred into the other beaker. This is what you would call capillary action. Capillary action is when water is able to squeeze into narrow spaces without the help of external forces (gravity). Capillary action can help with other things as well. Because it is able to transfer things, capillary action is important to trees and plants. It is important to trees and plants because that way, plants and trees are able to give each other nutrients that they need.
Hello again, world! How did you like it? I hope I was able to give you very important information that you will probably need later in your life. If you didn't need it later in your life or already knew it, it doesn't hurt to know a couple extra things, right? By saying that, I am done. I hope to see you reading from my website again. Peace! ; )



